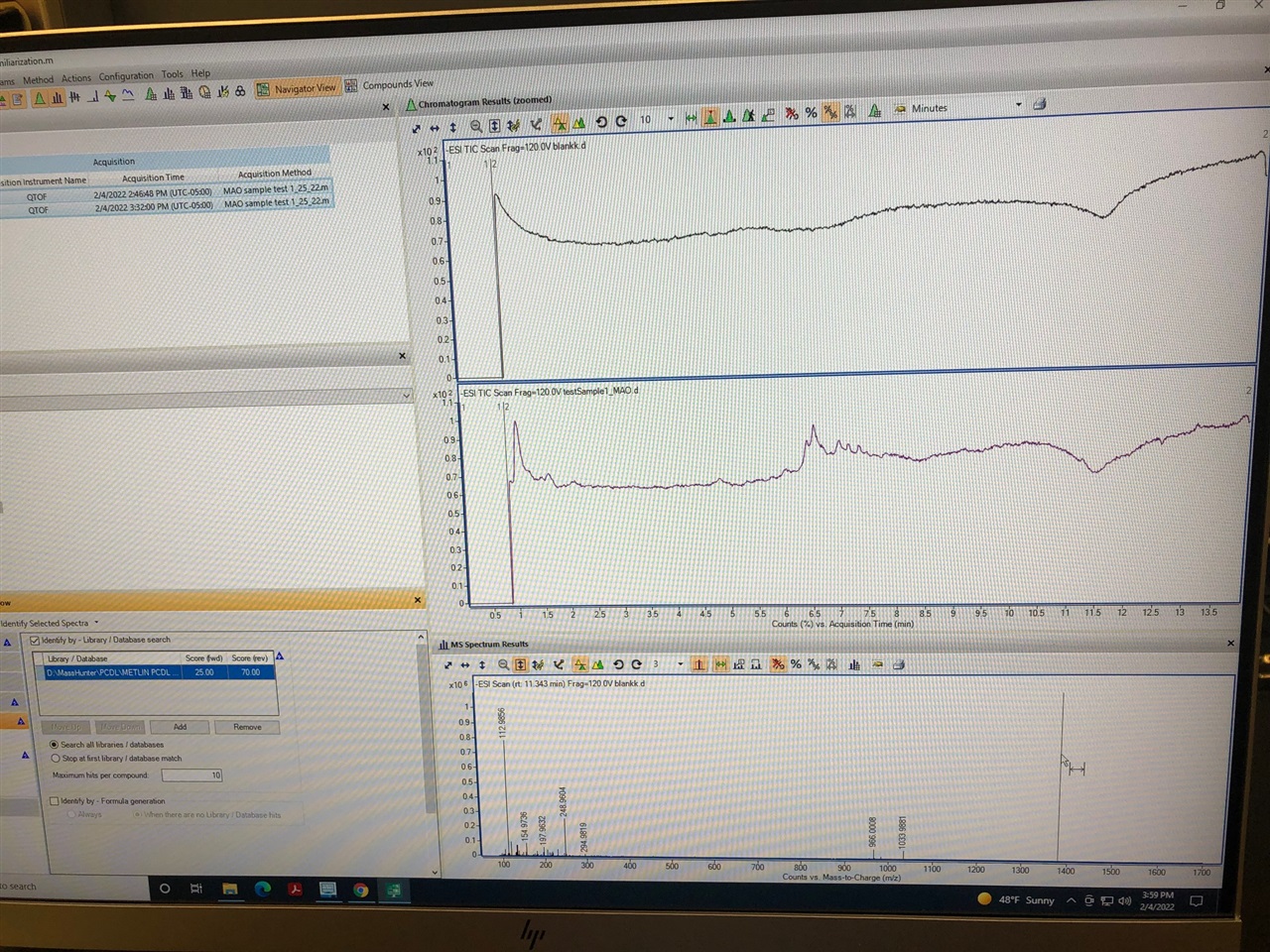
My chromatograms using 6545 LC MS runs above zero on x-axis. What can be the potential reason?

My chromatograms using 6545 LC MS runs above zero on x-axis. What can be the potential reason?
AHAS-
You can select the chromatogram by left-clicking on it. You can then right click and select "Adjust". It will adjust the selected chromatogram to the specified percent of full scale. The specified percent is set by right click in the Real Time Chromatogram window and selecting Change. The Y scale is also set there.
Thank you. I changed the offset % to zero but still TIC chromatograms starts above 1!
When I go to the MH qualitative analysis and click on Scale to Largest in All Chromatograms (the % sign) it shows from zero but when I Scale Chromatogram Off, it is not showing from zero. It is bothering me because it has been a month I am trying to figure this out (maybe it is just a simple thing to do and I am not aware of) but no luck! I have the older chromatograms with our old LC/QTOF in our lab and people were getting nice peaks and chromatograms with nice baselines starting from zero. With this new Agilent system it has never been like this..
This is normal on this type of instrument. The chromatogram you have displayed is a TIC Total ion chromatogram or Total ion count. It is the sum of all the ions of all m/z that are detected. In LCMS there will always be ions detected, these come from sample, solvent background and your reference ions. Because of this, and unlike a UV detector for example, we cannot zero or balance the MS at the start of a run. You won’t really analyze the TIC, typically you will extract chromatograms for the m/z of interest EICs Extracted ion chromatograms) or the use one of the algorithms to mine compounds from the data eg Find by formula for targeted analysis or find by molecular feature for untargeted analysis.
What you see in qual is the ion count or abundance with Scale Chromatogram Off. The Scale to Largest in All Chromatograms plots everything as a % of the highest abundance in all your data.
If you extract a BPC, base peak chromatogram it might be more informative.
If you know what compounds you are looking for use Find By Formula or extract EIC Ion extracted chromtograms of the exact mass of the ion to 4 places.
Thank you for your response and information. It makes sense, maybe sample background is dirty or solvent.
However, I have attached a TIC chromatogram from the old HPLC/QTOF in our lab and I like how the TIC chromatogram looks like. Then when I check for the EIC it shows up as nice sharp peak. When I look for EIC it shows nice peak too but I was just wondering how can I get nice TIC like as before with older instrument. Would it be the sensitivity for the new instrument that it picks up the background from sample or solvents? I want to mention that I ran this sample without reference ions checked and changed the solvents to the new bottles but not sure about the sample background? maybe need to filter or centrifuge more before run?
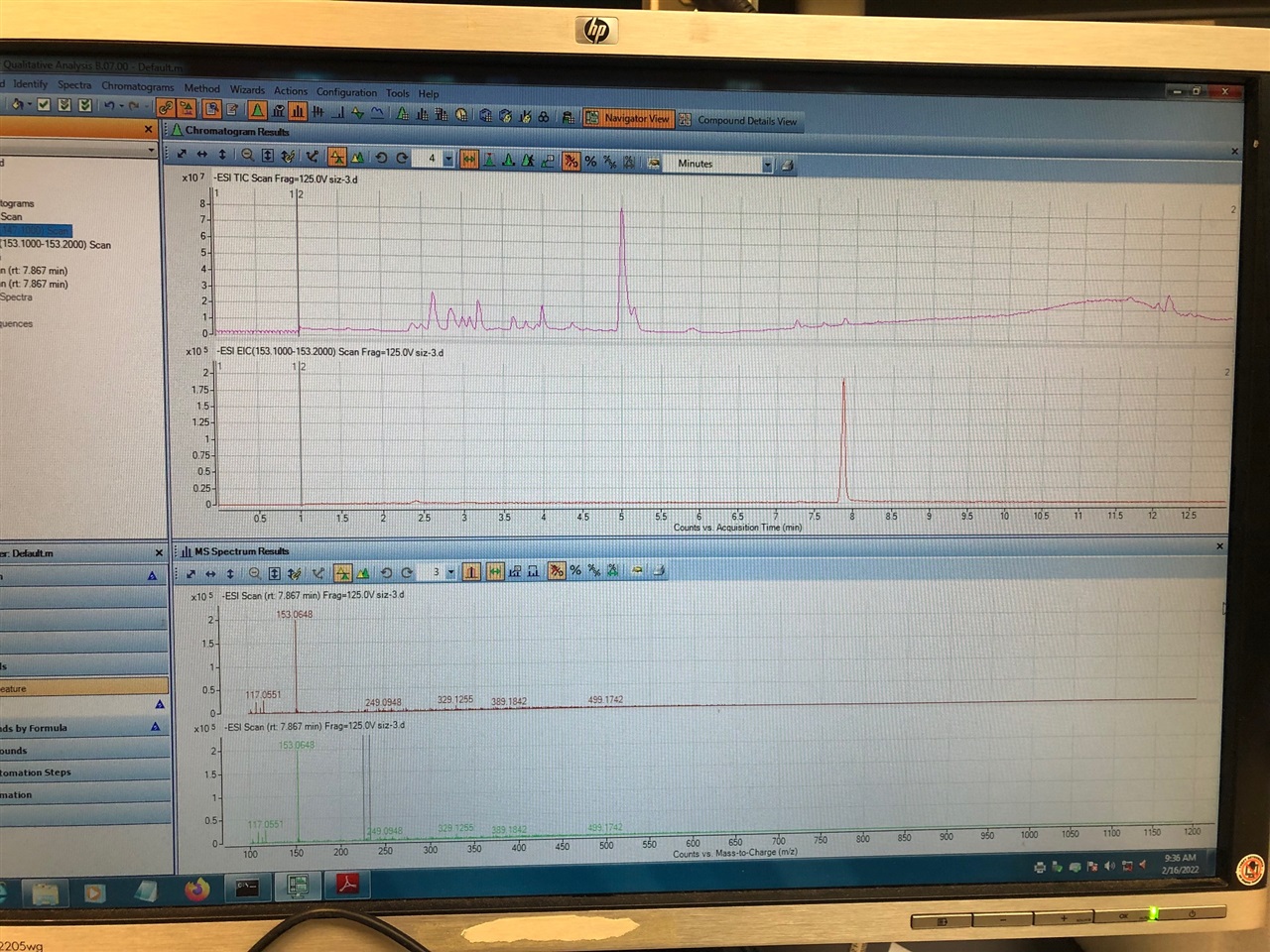
Look at the difference between the spectra in the background. It looks like the older system has a high background signal at m/z 112.9. I wonder if this system has been used with trifluoroacetic acid? That ion, in negative mode, might well correspond to residual TFA in the system!
Andy
I think you meant in the new system? The first chromatogram with high background is our new system. We have not used TFA in mobile phase or anything else, but added as reference solution, does it come from the reference?
Hi Has
I got the chromatograms the wrong way around, so you're right, I probably meant the new system. There should be no TFA in the reference solution.
/Andy
Thank you Andy for your feedback and time. I am not sure about TFA in the solvent bottles but never had this problem in the beginning. The thing is, I can see other ions high too like 134.86 not only TFA! In the lower mass there was not such high background before on -ve mode.
We had the older HPLC connected to the new MS system though. Do you think this might have caused the background issue? like all the contamination come to the new MS? We just hooked up the new HPLC to the MS couple days ago and I see all these background getting worse now.
Hi Has
Have you managed to overcome the high background? I would probably look at using the Agilent flushing solution on the HPLC, to make sure that this is properly clean, if you are still struggling with high background.
/Andy