Hi everyone : We use an ICPMS 7800 and, before and after the first standard calibration, the signal intensity vary a lot.
The intensity for most of the elements tracked decrease strangely, even if we take care of cleaning the autosampler probe with washes before and after the calibration.
- Matrix of Standards : 2.7% v/v HNO3
- Matrix of wash ("Rince pre-std", "Diluent Blank") : 2% HNO3, 0.5% HCl
As you can see on the screenshot below, the intensity for the same solution can vary from single to double. On the same note, the intensity of the wash before calibration decrease slowly for most elements tracked, even with 10 washes. As a result, all the blanks following are negative until the next recalibration.
(knowing that we leave the ICPMS warm up 20 minutes at its start, before doing the performance report, a tune of the instrument, adding the internal standard line and 10 minutes of wash again, so the ICPMS is turned on for approximately an hour before the "Rince Pre-Std". We wash it as well at the end of each analysis with a dedicated wash solution, according to Agilent advises.)
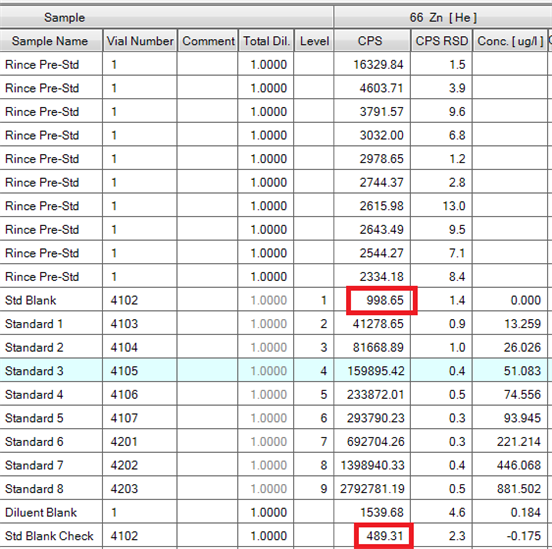
do you have any idea what could happen here? Thanks!
