Hi everyone :) I need your help !
As mentioned in the title, my question is about some kind of artefact on my chromatograms increasing as the column is used.
I will insert pictures at the end so it is clearer for you. (this message is quite long)
Here are some details :
Column : C18 ODS Hypersil
Mobile phase : Water acidified at pH 3,0 with phosphoric acid / Acetonitrile in proportions 99/1
HPLC : Agilent 1290 Infinity
Here is the context :
A solution of Product is injected. The goal is to measure content of Imp 1 in Component 1. On chromatogram we can see the peak of Imp1 at approx 4 min and a peak of the main component in my product at approx 8 min. Lactose monohydrate is an excipent. Therefore there is no peak corresponding to it on the chromatograms.
The more the column is used, the bigger the "artefact" gets. This artefact is interfering with the peak of Imp 1.
Sometimes the artefact is so big that I can't even see my Imp 1 peak.
I thought of several hypothesis but I still don't really understand what is going inside the column during the analysis :
- Proportions of aqueous phase in the mobile phase : 99 is a lot and can influence hydration of the stationary phase. But I don't observe Rt shift.
- Presence of lactose has in fact an effect but I don't know how it could interact with maybe C18 chains in the column and free silanols ?
Aspects of my chromatograms : normal aspect vs aspect when "artefact" has increased :
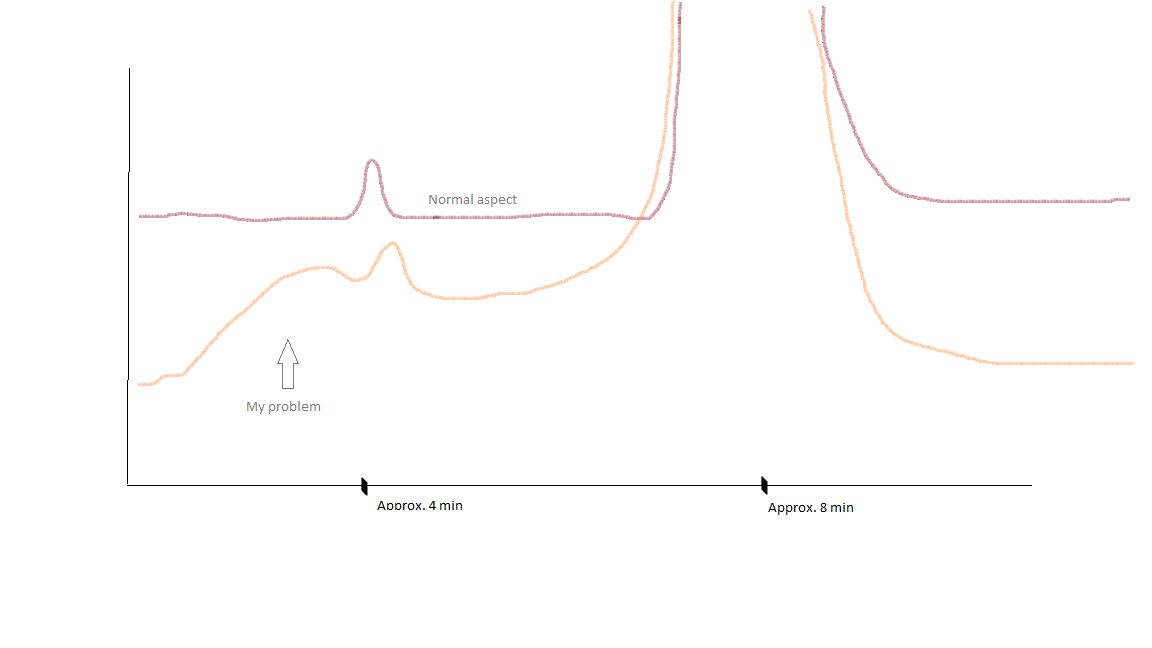
The artefact does not appear like that. It becomes bigger as the colums is used. But sometimes it appears sonner, I don't know how to explain it, it's not really repeatable.
Here is the forumula of my Imp 1 (Rt = approx 4 min) :
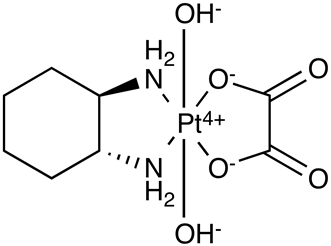
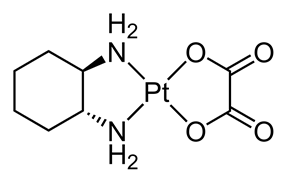
Here is the formula for information of the main component (Rt = approx 8 min) :
Sorry for this very long message,
Do you think you can help understand what can cause the artefact ?
Emeline
