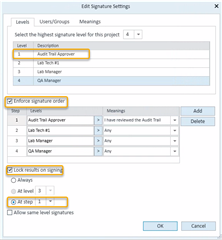
Is it possible to add a field to a report which indicates whether the audit trail was reviewed or not, and by which user?
I know that you can draw an audit trail report, but our audit trails can be 80 pages long! So if we can just add a field to one of our other reports that will indicate whether or not the audit trail for a results set was reviewed, then we won't need to print out the 80 page audit trail and can just keep a digital copy on file.